Why The Radiative Capabilities Of Gases Do Not Contribute To The Greenhouse Effect.
Abstract.
This essay is an attempt to link real world observations (the failure of surface temperatures to rise in tandem with atmospheric CO2) to basic physics and thereby show why the radiative characteristics of Greenhouse Gases cannot increase the surface temperature of a planet when atmospheric mass, the strength of the gravitational field and the power of insolation at the top of the atmosphere remain the same.
Only atmospheric volume and thus circulation can be affected.
The Basic Building Blocks.
For an atmosphere to develop one needs only three factors:
i) A spherical planetary body (usually rotating) large enough to generate a gravitational field strong enough to hold an atmosphere.
ii) Materials on the surface capable of forming a gas when irradiated.
iii) A suitable source of irradiation, inevitably a nearby sun.
Incoming radiation initially converts surface materials to gaseous form by heating them. In the process of forming, the new gases acquire energy from the surface by conduction. Until the gases reach their maximum possible temperature the surface will be a little cooler than it otherwise would have been.
Once established, the atmosphere then begins to pass energy to and receive energy from the surface in an endless cycle of balanced adiabatic ascent and descent driven by conduction at the surface causing density differentials in the air. On a rotating spherical world the surface heating is always uneven so a convective circulation is established carrying energy up and down, from equator to poles and back and from day side to night side and back.
The rotation spreads the temperature differentials latitudinally forming discrete climate zones with favoured regions of either ascending or descending air with jet streams threaded between them.
The proportion of incoming solar energy that the mass of the atmosphere can retain will determine the height above the surface to which its molecules can rise.
That height is dependent on a combination of the mass of the atmosphere and the strength of the gravitational field for any given level of incoming irradiation.
A stronger gravitational field will produce a lower, denser, warmer surface than a weaker gravitational field since the amount of solar energy retained by the atmosphere will be focused into a smaller volume and that amount of energy will be determined by the amount of mass available to absorb it at any given level of solar irradiation.
It is the amount of energy required to lift the bulk atmospheric mass off the surface and hold it off the surface that determines the temperature that will be attained over and above that required to match outgoing energy with incoming energy.
On Earth, that additional kinetic energy required to maintain the height of the atmosphere raises the temperature of the surface by about 33C above that expected from the S-B equation.
Greenhouse gases do absorb more energy than radiatively inert gases but they do not get any warmer. Instead, they simply rise to a higher level, converting kinetic energy to potential energy in the process until they reach a height at which they are at the same temperature as surrounding gases.
CO2 molecules are heavier than air and would be expected to be densest near the surface. However they are well mixed in the atmosphere and reach high levels due to their ability to absorb radiation combined with intense collisional activity in the denser lower regions.
Once thermal equilibrium has been reached between surface and atmosphere the surface will have become warm enough to both cycle energy between the surface and the atmosphere in perpetuity via conduction and convection AND have enough warmth left over to emit energy from the top of the atmosphere as fast as new energy comes in from the sun.
That is the reason why the surface must be warmer than the temperature predicted by the S-B equation and it is all about atmospheric mass held off a surface within a gravitational field and irradiated by an external energy source.
Retaining an Atmosphere.
For an atmosphere to be retained long term it is necessary for the amount of energy coming in from the sun to be matched by energy leaving the atmosphere for space.
Any permanent imbalance leads eventually to the loss of the atmosphere either by it freezing to the surface or being inflated so much that it drifts off to space.
Thus it is essential that if there are any factors that might disturb the top of atmosphere energy balance over the long term there must be negative feedbacks in place that are strong enough to offset those factors.
Such potentially disruptive factors would include composition changes (not involving changes in atmospheric mass) on the surface or in the atmosphere. That includes radiative absorption characteristics.
It is often suggested that GHGs alter the radiative flow of energy up through an atmosphere so as to allow warming at the surface and cooling higher up whilst not upsetting the top of atmosphere radiative balance permanently so that the lower atmosphere and surface settle down to a higher equilibrium temperature.
It is a fundamental tenet of anthropogenic global warming theory that all the warmth at a planetary surface above that predicted by the S-B equation is due to those GHGs rather than atmospheric mass.
I find that unlikely. If the surface and lower atmosphere become warmer than the temperature set by insolation, gravity and mass they will emit more energy to space, there will be more energy outgoing than incoming and the system will cool back to the earlier temperature.
I will now analyse how the system could work and show that composition changes not involving changes in mass only affect atmospheric volume and circulation patterns and not surface temperature. I will focus on composition changes involving greenhouse gases though other composition changes are possible.
The Radiatively Inert Atmosphere.
When there are no greenhouse gases the atmosphere cannot absorb incoming radiation or emit outgoing radiation. That has led some to propose that after a while such an atmosphere becomes ‘isothermal’ (the same temperature from top to bottom) at the same temperature as the surface save for a shallow layer in contact with the ground which can warm and cool conductively with the ground as sunlight comes and goes.
In such a scenario convection would stop and the surface would become no warmer than the temperature set by the S-B equation.
There would be little or no energy cycling between the mass bulk of the atmosphere and the surface so the surface need only be warm enough to provide the necessary radiation out at the top of the atmosphere.
The radiatively inert atmosphere would therefore not provide the so called greenhouse effect.
There is a flaw in that popular proposal.
Being a sphere, a planet has three dimensional geometry so that as one goes up away from the surface the amount of space available in the horizontal plane increases exponentially.
Consequently, the higher a molecule rises from the surface the more space it has all around it and the less pressure there is from above.
That reduction in density results in a temperature fall because the kinetic energy carried by the molecule when it started at the surface was gradually replaced by potential energy as it rose. The potential energy comes both from the lifting of the molecule away from the surface and from the widening separation of the molecule from other molecules at the same height.
Kinetic energy registers as heat but potential energy does not so there will still be cooling with height and vertical temperature differentials will cause a convective circulation to develop so as to prevent an isothermal structure forming.
To become cooler with height an atmosphere does not need to have radiative capability. It is sufficient for the gases to rise so that kinetic energy is converted to potential energy.
One could get an isothermal structure in a closed sided vertical container with a sealed top , a flat base and no rotation so perhaps we should call those who insist on an isothermal structure being possible ‘Flat Earthers’ ?
There is no net loss of energy as one goes up through an atmosphere because a molecule at the surface has the same total energy as a molecule at the top of an atmosphere. At the surface it carries 100% kinetic energy and at the boundary of space it is almost 100% potential energy. The potential energy never quite gets to 100% because space is not at absolute zero.
Space has been correctly described at a potential energy sink at the top of the atmosphere with a temperature of only 3k above absolute zero.
The concept of an isothermal atmosphere requires a bizarre proposition.
The energy content of such an atmosphere would be skewed towards the top with the molecules at the boundary of space containing both a full load of kinetic energy AND a similar amount of potential energy whereas those at the bottom would have kinetic energy only.
That would result in indefinite expansion at the top of such an atmosphere and it would be lost to space. The gravitational field would not be able to constrain such energetic molecules high up.
So, in fact a radiatively inert atmosphere will still have a convective circulation, there will still be an energy exchange cycling adiabatically between surface and atmosphere, the entire mass of the atmosphere will be involved and the surface will be warmer than the temperature predicted by the S-B equation.
All that will be achieved without greenhouse gases.
The slope of the temperature decline with height will be determined by the strength of the gravitational field acting on the entire mass of the atmosphere. That is the origin of the so called ‘lapse rate’ but as we shall see that slope and the heights to which it travels can be caused to vary as a result of compositional changes within an atmosphere such as the introduction of greenhouse gases. It is varying of the slopes and heights which changes the rate of non-radiative energy flows up from the surface and thereby negates the thermal effect of greenhouse gases.
The Thermal Behaviour of Greenhouse Gases
The radiative characteristics of greenhouse gases do not alter surface temperature but instead affect atmospheric volume and circulation.
In an atmosphere open to the sky as opposed to a container in a laboratory a density change is forced by the absorption of radiative energy by the GHG molecules. The consequent work done results in extra uplift with the affected GHG engaging in collisional activity with the surrounding non GHG molecules and the whole mixture rising vertically against the gravitational field to a higher level than would otherwise be the case.
That uplift causes all the additional radiative energy to be converted from kinetic energy (which registers on sensors as heat) to potential energy (which does not register on sensors as heat).
The warmed, higher molecules cool at a rate determined by the decline of pressure with height (the lapse rate). They will keep rising through collisional activity until they encounter an inversion where temperature starts to increase with height and on Earth that is the tropopause.
At that point the mixture of radiatively warmed molecules will settle at the same temperature as the surrounding molecules already at the new height and join the general circulation. All the extra radiative energy added will have gone from Kinetic energy to Potential energy at the new height leaving no energy left over for an increase in surface temperature.
The failure to raise surface temperature whilst increasing atmospheric height is due to the physical effect of the Gas Constant and I will deal with that in more detail in a separate essay. The Gas Constant is the ultimate global thermostat.
Molecules with radiative absorption capability will produce an atmosphere of a different volume at the same surface temperature and mass as compared to an atmosphere without such a capability.
That difference in volume will be over and above any difference in volume arising from differing molecular weights of the constituent gases.
That additional radiatively induced difference in volume can only alter the proportions of kinetic and potential energy. It cannot affect surface temperature as well. The Gas Constant being based solely on mass will not allow it.
Not all GHGs behave the same way due to other variable physical characteristics. By way of illustration I will describe the effects of one non -condensing and one condensing GHG.
Add a condensing GHG
I will consider only water vapour here because it is so important for the thermal stability of Earth’s climate system.
Water vapour is lighter than air once formed by the acquisition of the latent heat of evaporation so no additional radiative energy needs to be acquired to enable it to rise within the Earth’s gravitational field. No increase of surface temperature is needed to make it rise to a level where the lapse rate makes it cooler.
Although water vapour is a greenhouse gas it had no warming effect at the surface where the vapour simply acquired the same temperature as the surrounding air molecules.
The vapour, being lighter than air then rises, in the process altering the lapse rate from the dry rate to the moist rate. At the dew point clouds condense out and the latent heat of condensation releases more energy which drives the convection higher. Deeper denser clouds prevent some solar irradiation reaching the surface by reflecting it back out to space.
The vapour has then gone, falling out as rain or floating around as cloud. It does not participate in the subsequent adiabatic descent cycle. It is a one way upward escalator moving surface energy faster to space.
Although water vapour is a greenhouse gas its effect is overwhelmingly one of surface cooling.
For water vapour to have a surface warming effect it would need to remain at the surface absorbing more energy and rising in temperature until uplift could start. That does not happen.
It is a greenhouse gas but its thermal effect is entirely negative due to it being lighter than air from its creation.
Add a non condensing GHG.
I will consider only CO2 since that is the cause of most concern.
CO2 is heavier than air and so needs to acquire additional energy to make it rise.
However the radiative absorption capability of CO2 is also high so in practice it has little difficulty acquiring the energy needed to initiate uplift.
Furthermore, it can conduct energy via collisional activity to nearby non -radiative molecules encouraging them to rise too.
When CO2 molecules (or any other GHGs) absorb more energy than those around them their higher temperature increases the spaces between them and adjoining molecules reducing density and causing them to rise until they reach a height where they are at the same temperature as surrounding molecules.
Note that the mass (m) of the atmosphere remains the same, surface pressure (P) remains the same at a given strength of gravitational field whatever the volume of an atmosphere and kinetic energy ( T) for the atmosphere as a whole stays the same because no additional energy is being added to the system from outside.
If T were to be permitted to rise then the surface would be too warm for top of atmosphere energy balance plus the cost of maintaining atmospheric height,
Too much energy would radiate out and the temperature would fall back to a lower level.
In the process of rising higher all of any additional energy absorbed radiatively by CO2 is simply converted to a change in potential energy content.
The logical reason for that is that once the surface is warm enough to support both the mass of the atmosphere and to emit as much energy out as is received then any extra kinetic energy is not required for maintaining the system so it does work instead and that work is the lifting of the affected molecules higher so it goes straight to potential energy and not kinetic energy (T).
All of the ‘surplus’ kinetic energy is converted to potential energy with nothing left to raise surface temperature.
Summary and conclusions.
This essay has tried to show that the concept of greenhouse gases contributing to the mass and gravity induced ‘greenhouse effect’ is wrong.
All the so called greenhouse effect amounting to some 33C is a result of mass and gravity alone.
Evidence in support includes:
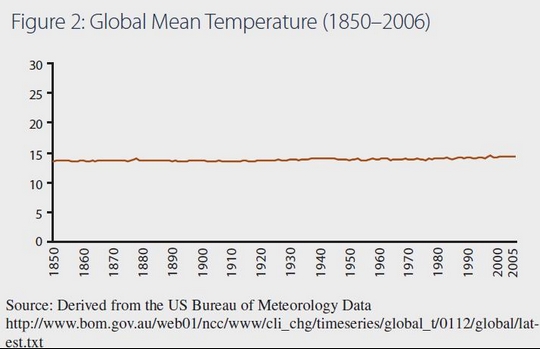
i) The current pause in surface warming despite apparently much increased atmospheric CO2
ii) The absence of the so called ‘tropospheric hot spot’ which would have developed if the atmosphere had failed to expand appropriately in response to the composition change.
iii) The failure of outgoing longwave radiation (OLR) to show that there was any increased atmospheric blocking of outgoing radiation following short term troposphere warming episodes.
iv) the failure of the oceans to become any warmer since the deployment of Argo sensors about ten years ago.
Such observations negate the CO2 based theory but are entirely consistent with the proposals set out here.
The radiative absorption capability of CO2 allows atmospheric molecules to reach a higher temperature than that imparted to them by energy at the surface so they rise to a higher location than would be predicted from their weight and their individual gas constants.
CO2 molecules do not rise singly on their own because they conduct some of their extra energy to surrounding non GHGs and the whole parcel of air rises higher with kinetic energy being converted to potential energy to a greater extent than for a radiatively inert atmosphere.
Given that CO2 is heavier than air we would expect it to be mostly at the surface but in fact it is a ‘well mixed’ gas in terms of height and there is plenty high up in the atmosphere. Their observed height and mixing ability is a result of radiative characteristics supplementing the energy they acquire from the surface and that additional energy being diffused through the whole atmosphere by collisional activity.
Additionally, GHGs are capable of radiating directly out to space which is an ability almost completely lacking in Nitrogen, Oxygen and Hydrogen. The greater height of GHGs would therefore facilitate extra radiative energy loss to space.
That would explain the absence of a tropospheric ‘hot spot’ since the energy that was supposed to accumulate higher up would simply have been radiated out directly to space from a greater height.
Radiatively warmed (whether directly or indirectly through collisions) molecules are placed higher in the atmospheric column than can be explained just from their individual gas constants and once at that height have an enhanced cooling effect equal to their enhanced warming effect with a zero net effect on surface temperature.
Part of the enhanced cooling will be from direct radiation out from the atmosphere but the bulk will be from conversion of kinetic energy to potential energy in the expanded atmosphere.
As a separate issue CO2 molecules would have an imperceptible effect on surface temperature anyway because mass and gravity provide almost all of the greenhouse effect and CO2 comprises a miniscule proportion of the atmospheric mass.
Then we must consider the effects of sun and oceans on climate variations.
It is becoming increasingly clear that even if everything else I suggest is wrong we would still not be able to identify the tiny climate effect of our emissions as compared to that from solar and oceanic variability.
Published by Stephen Wilde October 26, 2013